ISO 13485 Risk Management Plan
In the intricate world of medical device production, where precision and safety are paramount, the ISO 13485 Risk Management Plan emerges as a beacon of assurance and meticulous planning. This plan template, identified by the number 8022, is not just a document but a comprehensive framework designed to navigate the complex landscape of risks inherent in the development and manufacturing of medical devices.
At its core, the ISO 13485 Risk Management Plan is a meticulously crafted blueprint that guides organizations through the labyrinth of potential hazards, ensuring that every conceivable risk is identified, assessed, and mitigated. It is tailored to align with the stringent requirements of ISO 13485, the international standard for quality management systems in the medical device industry. This alignment ensures that the plan is not only robust but also compliant with global regulatory expectations, providing a seamless pathway to market approval and patient safety.
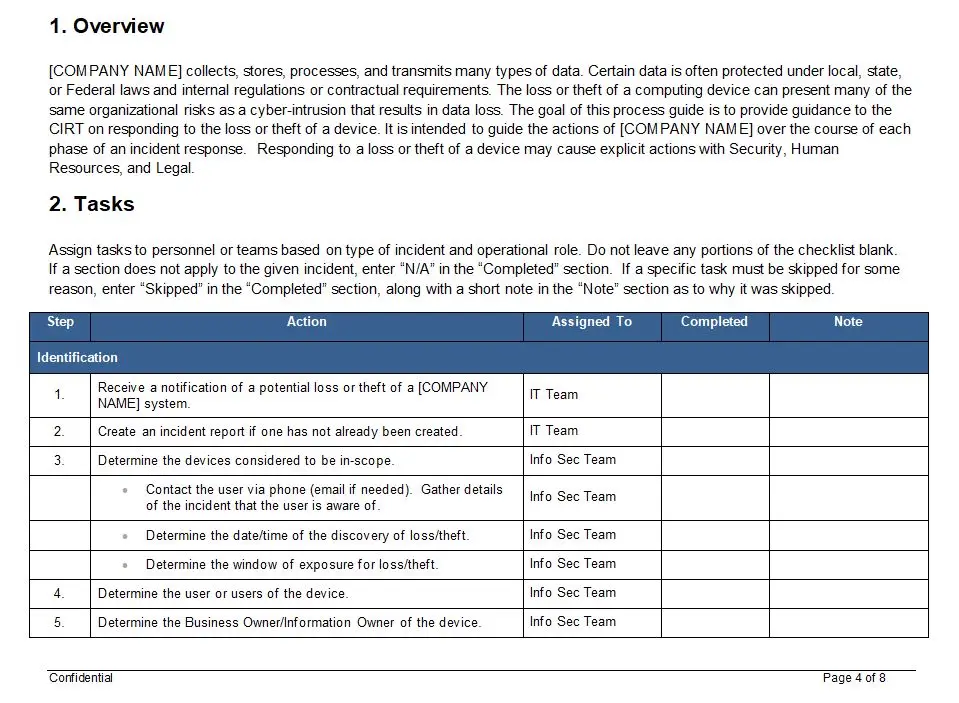
One of the key features of this plan is its structured approach to risk management. It offers a step-by-step methodology that begins with risk identification, where potential threats to product quality and patient safety are systematically cataloged. This is followed by a thorough risk analysis, where each identified risk is evaluated for its severity and likelihood, allowing organizations to prioritize their risk mitigation efforts effectively.
The plan also emphasizes the importance of risk control, providing strategies and tools to reduce or eliminate risks to acceptable levels. This proactive approach not only safeguards the end-users but also enhances the reliability and performance of the medical devices, fostering trust and confidence among healthcare professionals and patients alike.
Moreover, the ISO 13485 Risk Management Plan is designed to be dynamic and adaptable. It encourages continuous monitoring and review of risks throughout the product lifecycle, ensuring that new risks are promptly addressed and that the risk management process evolves in response to changes in technology, regulations, and market conditions. This adaptability is crucial in an industry characterized by rapid innovation and stringent regulatory scrutiny.
The benefits of implementing the ISO 13485 Risk Management Plan are manifold. For manufacturers, it serves as a critical tool for minimizing product recalls and liability issues, thereby protecting the company’s reputation and financial stability. For regulatory bodies, it provides assurance that the manufacturer is committed to maintaining the highest standards of quality and safety. And for patients, it offers peace of mind, knowing that the devices they rely on are produced with the utmost care and diligence.
In essence, the ISO 13485 Risk Management Plan is more than just a template; it is a strategic asset that empowers organizations to navigate the complexities of medical device production with confidence and precision. By embedding risk management into the very fabric of their operations, companies can not only achieve compliance but also drive innovation and excellence in the pursuit of safer, more effective medical solutions.
All GovernanaceDocs documents are developed based on well-known standards such as NIST CSF, ISO 27001, ISO 22301, PCI-DSS and HIPAA.
Hence, You just need to download and selected document and add your company name and logo.








Reviews
There are no reviews yet